Overview
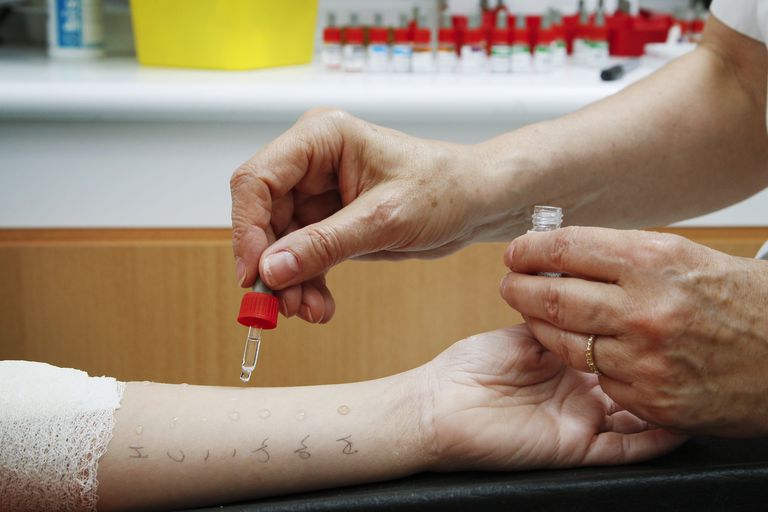
Allergy Skin (Prick) Test
It is a simple, pain-free and safe diagnostic method that cause minimal complication and can yield reports very quickly to identify substances, which lead to allergy. This test helps determination of strategies to protect the patient against the relevant allergy by identify the allergens that lead to complaints and cause the allergic reaction. The test is also used to evaluate treatment outcomes in patients who receive immunotherapy. Prick test is a commercially available test that uses respiratory and food allergens, latex or (rarely) drugs, which may cause allergic reaction in people. It is used for allergic diagnosis of respiratory and non-respiratory allergens.
Test procedure: Ready-to-use allergens are injected to medial surface or dorsum of forearm using special needles. The test evaluates redness and bumps that develop 15 to 20 minutes after allergens are injected at the intended site.
Interpretation of Results: Redness and bumps are measured in millimeter and score from zero to four positive. Allergic substances are identified. Results are reported to patients within 30 minutes.
Immunotherapy (Vaccination Therapy) in Allergy
Our body is continuously exposed to external and internal viable and non-viable substances. Some of them are hazardous under normal circumstances (allergens), while others cause serious hazards (bacteria, viruses, fungi etc.). However, below mentioned factors may join and cause allergic diseases.
- Genetic factors or familial predisposition
- Specific allergic factors (dust, pollen, foods, drugs etc.)
- Non-specific environmental factors:
- Smoking
- Infections
- Exercise
- Psychological disorders
- Various therapies (radiotherapy, chemotherapy etc.)
These factors join over time and cause allergic diseases.
Immunotherapy has been used for a very long time and it aims training the immune system to teach that allergens are not enemies for the body (to boost tolerance). The allergic substances are administered at gradually increased doses to modify the immune response over a long period of time and thus, no reaction develops in the next encounter with the allergen.
Most of drugs used in medical treatment of allergy suppress most symptoms for a long term, but they do not cure the disease. Immunotherapy is still the only treatment modality that is successfully performed to eliminate the disease.
Who are good candidates for immunotherapy?
When patients are selected for immunotherapy, allergens that cause secretion of specific IgE (a specific antibody released against the allergen) should be identified and it should be evidenced that clinical symptoms aggravate while exposure to the allergene.
Immunotherapy can be used as a preventive measure;
- For allergies that cannot be controlled despite allergen prevention methods and use of medications,
- Inability to use the medication due to side effects of anti-allergic drugs,
- To prevent development of new allergies and asthma.
What are contraindications of immunotherapy?
- Severe and uncontrolled asthma,
- Severe immunodeficiency,
- Cancer patients,
- Severe heart diseases,
- Use of beta-blocker agents,
- Incompliance of the patient,
- Children younger than 6,
- Pregnancy (recent)
Immunotherapy is not recommended for above mentioned conditions. Moreover, immunotherapy is not performed in food allergy, eczema and hives.
If it is started before immunization in pregnant women and the patient tolerates the treatment, immunization can be maintained throughout the pregnancy, but a new immunization protocol is not started in the pregnancy. If the person conceives while the dose is adjusted, it is not increased and the patient is switched to maintenance treatment at the maximal dose administered. Spontaneous miscarriage, premature delivery or neonatal hypoxia is likely, if systemic reaction occurs secondary to allergy vaccines administered in pregnancy. Immunotherapy can be safely administered and mostly tolerated well in children. However, incompliance problems can be experienced especially in immunization at regular intervals in children. Although a hazard of immunotherapy for patients with immunodeficiency or cancer has not been evidenced, these studies are, unfortunately, insufficient.
Immunotherapy is given in two phases; baseline and maintenance.
At baseline phase, allergens are diluted by special solutions and administered to the body at regular intervals in very small doses that cannot trigger an allergic reaction. Each dose is administered once a week for several months in injection therapy or until maintenance dose is started in drop and tablet therapies.
Maintenance phase is started when the highest and most efficient dose is reached that does not cause a systemic reaction and is tolerated by the patient. The interval between drops and immunization is reduced. The treatment lasts for 3 to 5 years in average. Studies demonstrate that a 3- to 5-year immunotherapy offers efficiency, which lasts for many years, in patients with allergic rhinitis. It is revealed out that immunotherapy is a protective measure for patients with allergic rhinitis against development of asthma in the future.
Despite interpersonal variations, symptoms can be completely eliminated in some patients at the end of the treatment, while there may be minimal or no treatment response in others. It may take 6 months to 1 year to get a treatment response. It may be appropriate to stop treatment, if complaints do not regress at the end of first year.
It is recommended to take antiallergic medications throughout the immunotherapy until symptoms regress. Dose of these drugs is gradually decreased thereafter.
Subcutaneous Immunotherapy (Subcutaneous Vaccine)
The allergen solution is administered into subcutaneous adipose tissue at gradually increased doses using syringes with very thin needles according to a certain treatment schedule. The solution is injected to upper-lateral part of the arm, one arm in each injection. Since systemic side effects most commonly begin in the first half an hour, patients should be closely observed in the first half an hour after patients are vaccinated.
Minor local reactions that are in the form of redness or swelling may be seen at the injection site. Such local reactions do not require cessation of the treatment, but it may be necessary to adjust the doses depending on the severity. Bumps measuring up to 2 cm and persisting for 2 days are common, but they are insignificant. Anti-allergic medications and cold compression may help the larger ones. There is no evidence that such reactions progress into major systemic reactions.
Small nodules (palpable solid bums) can be seen at the injection site. They are not hazardous and do not require cessation of the treatment.
Although late reactions, such as headache and nasal congestion, can be seen, you need to consult your doctor, if any one of them is severe.
Severe systemic side effects of this treatment modality include generalized skin rashes, swelling and spasm in airways, vomiting, hypotension (low blood pressure) and shock.
Systemic reactions are more common at baseline phase, while the dose is adjusted. Since incorrect doses may also cause side effects, the dose and immunization schedule is checked twice by the doctor and the patient in order to follow up the treatment very carefully.
You should vocally check out the vaccine vial, the dose and administration date jointly by your doctor. This type of vaccines should necessarily by administered by healthcare professionals at healthcare facilities.
The health status of the patient should be checked before the injection. Injection is not recommended in case of common cold, lung diseases or other infections. Pulmonary functions should be tested in patients with asthma before the immunization. In case of systemic reaction, your doctor may consider to stop the treatment or reduce the doce.
Sublingual Immunotherapy (Sublingual Drop Vaccine)
On the contrary to the subcutaneous vaccine, it is administered by the patient at home and observation in a healthcare facility is not required. Allergen solutions are dropped beneath the tongue and swallowed after 2 minutes elapse. Drops are used every day at the baseline phase, while they are dropped three days of week in the maintenance phase. Adverse effects usually include itchy mouth cavity, swelling, nausea, abdominal pain and diarrhea. Generalized rash, asthma and anaphylactic shock are very rare, but no fatal reaction following sublingual immunotherapy is reported.
Tablet Immunotherapy
Oral tablet immunotherapy is one of the recent advancements in immunotherapy protocols. Allergens are administered every day in the form of tablets that dissolve quickly beneath the tongue. Side effect profile is similar to the sublingual immunotherapy. Tablet is placed beneath the tongue and kept in the cavity for 1 minute; it is swallowed after it dissolves. It is necessary to avoid eating and drinking for 5 minutes after the tablet is taken. These preparations are licensed for pollen mixtures in Europe and the U.S., while the forms for mites are under development. Pollen tablets are also licensed in our country.
Immunotherapy may lead to severe systemic allergic reactions that may threaten life. Therefore, they should be prescribed by physicians who are specialized in this field of medicine and allergens should be administered absolutely under supervision of a trained doctor in healthcare facilities that have adequate equipment for emergency procedures.





